Figure 1) Differences in the Atom Distances Between MOL-TALOU Model and Measured Values and Between Conventional Model and Measured Values
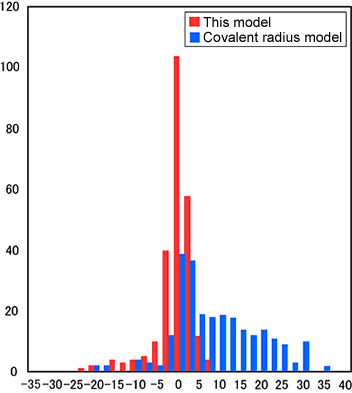
| X-axis: |
Number by type of bond |
| Y-axis: |
(Model-calculated value
-Measured value)/Measured value (%) |
|
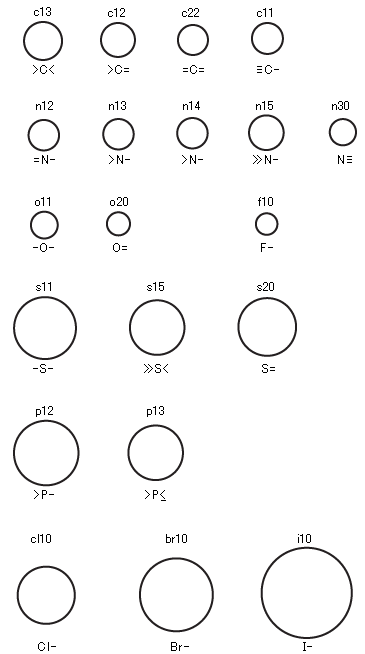
Figure 2) Relative Radii of Various Atom Balls
[How
to Obtain Actual Molecular Size]
Measure the molecular model in mm. If the measured value is given by d mm, the
actual molecular size can be obtained through the following formula:
d x 8/100 (A) or d x 8/1000 (nm) |
| [Model
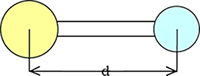
Measuring Method] |
| Measure the distance between the
centers of atom balls. |
 |
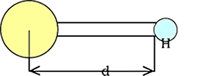 As an exception, the
distance between a hydrogen atom and another atom is measured from the tip of
the cylinder in the single hydrogen bond, not from the center of the hydrogen
ball.
As an exception, the
distance between a hydrogen atom and another atom is measured from the tip of
the cylinder in the single hydrogen bond, not from the center of the hydrogen
ball.
(This is because the radius of the hydrogen ball is considered 0 in a MOL-TALOU
model.) |
|